Fabricio S R Inoue
Conference 2022 Poster Presentation
Project title
3,3’,5,5’-tetramethoxybiphenyl-4,4’-diol promotes cell cycle arrest and apoptosis in hepatocellular carcinoma HepG2/C3A cell line
Authors and Affiliations
Fabricio Seidy Ribeiro Inoue1, Virginia Marcia Concato1, Bruna Taciane da Silva Bortoleti1,2, Ellen Mayara Souza Cruz1, Mariana Barbosa Detoni1, Fernanda Tomiotto-Pellissier1,2, Manoela Daiele Gonçalves3, Juliana Maria Bitencourt de Morais1, Elaine da Silva Siqueira1, Jéseka Gabriela Schirmann3, Aneli de Melo Barbosa-Dekker3, Robert Frans Huibert Dekker3, Ivete Conchon-Costa1, Idessania Nazareth Costa1, Mario Sergio Mantovani4, Danielle Lazarin Bidóia1, Wander Rogerio Pavanelli1
1. Department of General Pathology, State University of Londrina, Londrina, Brazil.
2. Graduate Program in Biosciences and Biotechnology, Carlos Chagas Institute (ICC), Fiocruz, Curitiba, Brazil.
3. Department of Chemistry, State University of Londrina, Londrina, Brazil.
4. Department of General Biology, State University of Londrina, Londrina, Brazil.
Abstract
Background
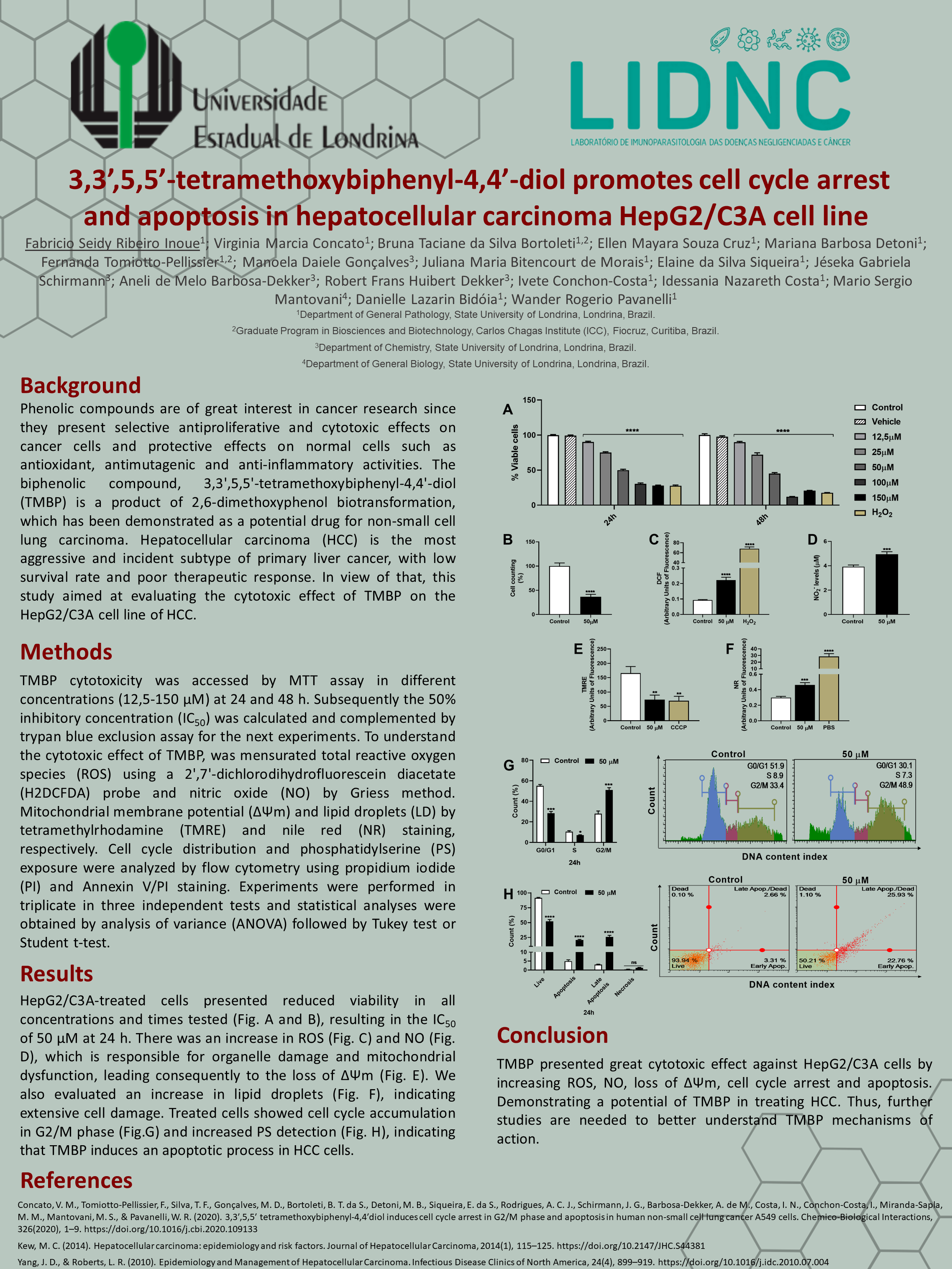
Phenolic compounds are of great interest in cancer research since they present selective antiproliferative and cytotoxic effects on cancer cells and protective effects on normal cells such as antioxidant, antimutagenic and anti-inflammatory activities. The biphenolic compound, 3,3′,5,5′-tetramethoxybiphenyl-4,4′-diol (TMBP) is a product of 2,6-dimethoxyphenol biotransformation, which has been demonstrated as a potential drug for non-small cell lung carcinoma. Hepatocellular carcinoma (HCC) is the most aggressive and incident subtype of primary liver cancer, with low survival rate and poor therapeutic response. In view of that, this study aimed at evaluating the cytotoxic effect of TMBP on the HepG2/C3A cell line of HCC.
Methods
TMBP cytotoxicity was accessed by MTT assay in different concentrations (12,5-150 µM) at 24 and 48 h. Subsequently the 50% inhibitory concentration (IC50) was calculated and complemented by trypan blue exclusion assay for the next experiments. To understand the cytotoxic effect of TMBP, was mensurated total reactive oxygen species (ROS) using a 2′,7′-dichlorodihydrofluorescein diacetate (H2DCFDA) probe and nitric oxide (NO) by Griess method. Mitochondrial membrane potential (ΔΨm) and lipid droplets (LD) by tetramethylrhodamine (TMRE) and nile red (NR) staining, respectively. Cell cycle distribution and phosphatidylserine (PS) exposure were analyzed by flow cytometry using propidium iodide (PI) and Annexin V/PI staining. Experiments were performed in triplicate in three independent tests and statistical analyses were obtained by analysis of variance (ANOVA) followed by Tukey test or Student t-test.
Results
HepG2/C3A-treated cells presented reduced viability in all concentrations and times tested, resulting in the IC50 of 50 µM at 24 h. There was an increase in ROS and NO, which is responsible for organelle damage and mitochondrial dysfunction, leading consequently to the loss of ΔΨm. We also evaluated an increase in lipid droplets, indicating extensive cell damage. Treated cells showed cell cycle accumulation in G2/M phase and increased PS detection, indicating that TMBP induces an apoptotic process in HCC cells.
Conclusions
TMBP presented great cytotoxic effect against HepG2/C3A cells by increasing ROS, NO, loss of ΔΨm, cell cycle arrest and apoptosis. Demonstrating a potential of TMBP in treating HCC. Thus, further studies are needed to better understand TMBP mechanisms of action.